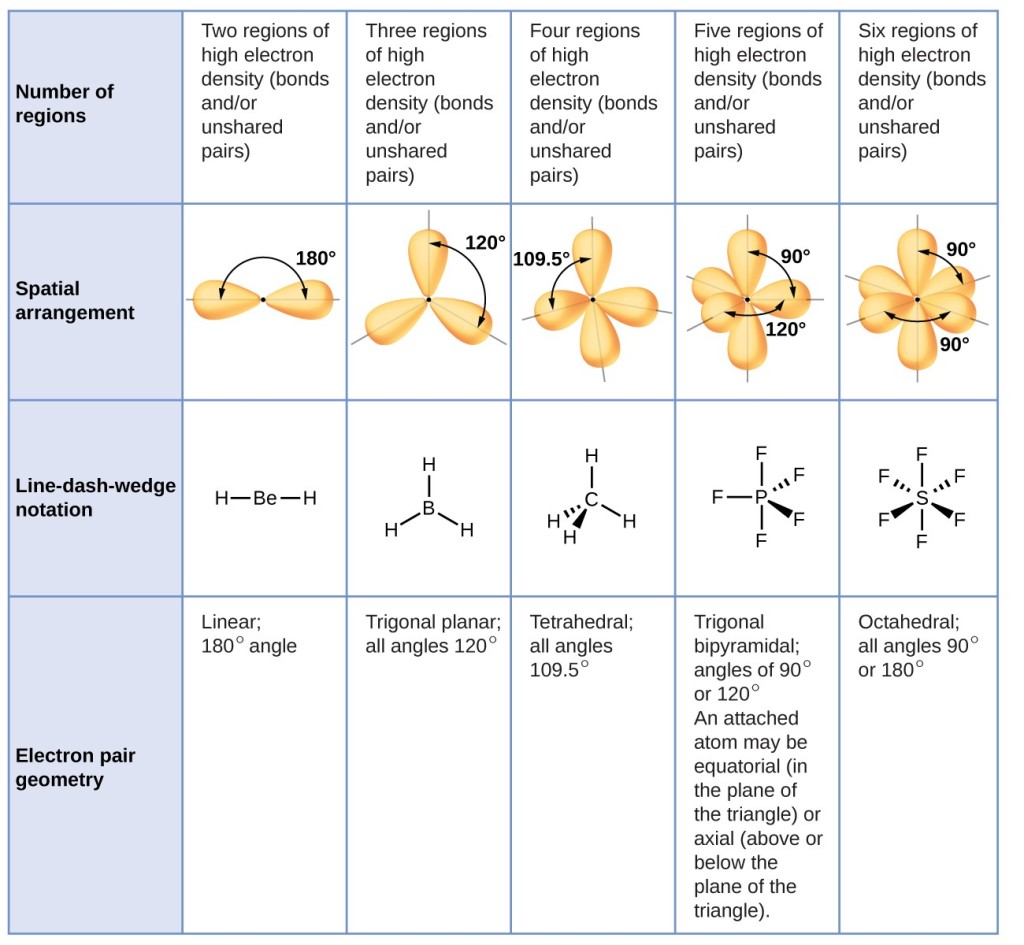
Your answers should be in terms of principles of molecular structure. Use the information in the table below to respond to the statements and questions that follow. (d) On the basis of your Lewis electron-dot diagram(s) in part (c), identify the hybridization of the sulfur atom in the SO2 molecule. Explain this observation, supporting your explanation by drawing in the box below a Lewis electron-dot diagram (or diagrams) for the SO2 molecule. (c) In the SO2 molecule, both of the bonds between sulfur and oxygen have the same length. The geometry of the regions of electron density is roughly the same as what we see when no NB pairs are involved. (b) On the basis of the Lewis electron-dot diagram that you drew in part (a), predict the molecular geometry of the IF3 molecule. Molecules with 1 NB pairs and only single bonds. (a) In the box provided, draw a complete Lewis electron-dot diagram for the IF3 molecule. The following questions are from past AP Chemistry exams that were posted online by College Board. The same principle applies to the other hybridizations. In the above example, carbon's 2p and 2s orbitals fuse into 4 half-filled sp3 orbitals that can make 4 sp3-orbital sigma bonds. Hybridization - You only have to memorize the hybridization of families 2, 3, and 4□. Shape - This is the main column that you should memorize and learn to associate with the general formula, electron domain geography, and hybridization. The graph below also includes angle measures that you should be aware of in that specific molecular geometry. It shows where the electrons or atoms are in relation to the middle atom, M. General Formula - made up of three parts:Įlectron Domain Geography - gives you an idea of what the molecule looks like. Once you practice, the questions that involve the VSEPR Theory become free points□!įamily - think of family as how many groups of atoms or molecules branch off the middle atom (number of x + number of e in the general formula). It gives you everything you need to know about VSEPR and will answer a lot of questions that require memorization on the AP. You should definitely memorize the table below for the AP Exam. It specifically uses the Coulombic repulsion between electrons as a basis for predicting electron arrangement. The Valence-Shell-Electron-Pair-Repulsion (VSEPR) theory can predict molecular geometry by minimizing electron-electron repulsion. Lewis structures can determine properties such as geometry, bond orders, bond lengths, and dipoles for molecules. In sp hybridization, one s orbital and one p orbital hybridize to form two sp orbitals, each consisting of 50 s character and 50 p character. Like an atomic orbital, a molecular orbital is full when it contains two electrons with opposite spin.Valence Shell Electron Pair Repulsion (VSEPR) The region of space in which a valence electron in a molecule is likely to be found is called a molecular orbital ( Ψ 2). Just like electrons around isolated atoms, electrons around atoms in molecules are limited to discrete (quantized) energies. 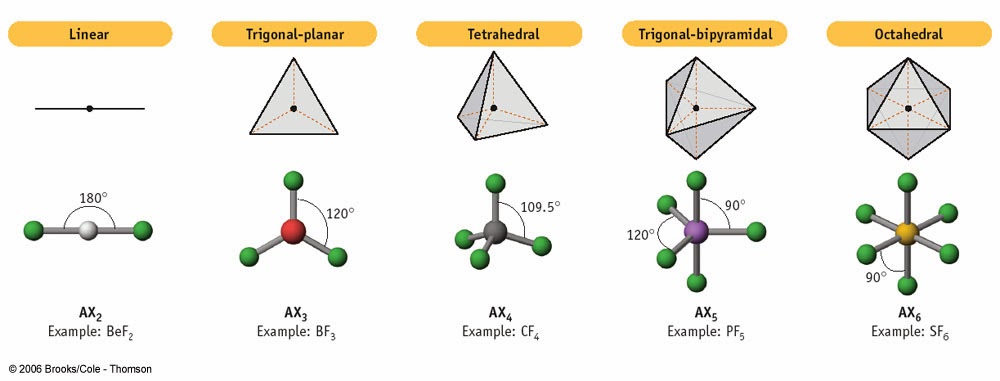
Using quantum mechanics, the behavior of an electron in a molecule is still described by a wave function, Ψ, analogous to the behavior in an atom. Molecular orbital theory describes the distribution of electrons in molecules in much the same way that the distribution of electrons in atoms is described using atomic orbitals. Needs multiple structures to describe resonance Predicts the arrangement of electrons in molecules Predicts molecular shape based on the number of regions of electron density Iodine pentafluoride (IF 5) is a polar molecule with net 4.81 D. The F-I-F bond angle in IF 5 is 81.9° while the I-F bond lengths are 184.4 pm and 186.9 pm. The central I atom in the IF 5 molecule is sp 3 d 2. \): Comparison of Bonding Theories Valence Bond TheoryĬonsiders bonds as localized between one pair of atomsĬonsiders electrons delocalized throughout the entire moleculeĬreates bonds from overlap of atomic orbitals ( s, p, d…) and hybrid orbitals ( sp, sp 2, sp 3…)Ĭombines atomic orbitals to form molecular orbitals (σ, σ*, π, π*)Ĭreates bonding and antibonding interactions based on which orbitals are filled The molecular geometry or shape of IF 5 is square pyramidal while its ideal electron geometry is octahedral.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
